The Science of Radon: From Discovery to Present Day
Discovery of Radon
Radon was first identified in the late 19th century during the study of radioactive elements. In 1899, physicists Ernest Rutherford and Robert B. Owens discovered a radioactive gas emitted from thorium, which they termed “emanation.” Shortly after, in 1900, German chemist Friedrich Ernst Dorn observed a similar phenomenon with radium and called the gas “radium emanation.” It wasn’t until 1908 that Sir William Ramsay and Robert Whytlaw-Gray isolated this gas and determined its properties, naming it “niton” (from the Latin word for shining, “nitens”). By 1923, the gas was officially named radon, aligning with the nomenclature of other noble gases and marking a pivotal moment in the history of indoor air quality research.
Creation of Radon Gas
Radon gas (Rn) is a noble gas, specifically the heaviest and the only radioactive one in its group. It is produced through the radioactive decay of radium-226, a decay product of uranium-238, found ubiquitously in soil and rocks. This decay sequence can be summarized as:
Uranium-238 decays to thorium-234, emitting alpha particles.
Thorium-234 decays to protactinium-234, emitting beta particles.
Protactinium-234 decays to uranium-234, emitting beta particles.
Uranium-234 decays to thorium-230, emitting alpha particles.
Thorium-230 decays to radium-226, emitting alpha particles.
Radium-226 decays to radon-222, emitting alpha particles.
Radon-222 has a half-life of about 3.8 days, after which it decays into a series of short-lived radioactive progeny, ultimately forming stable lead-206. Understanding this decay process is essential for radon testing and identifying long-term lung cancer risks in homes and commercial buildings.
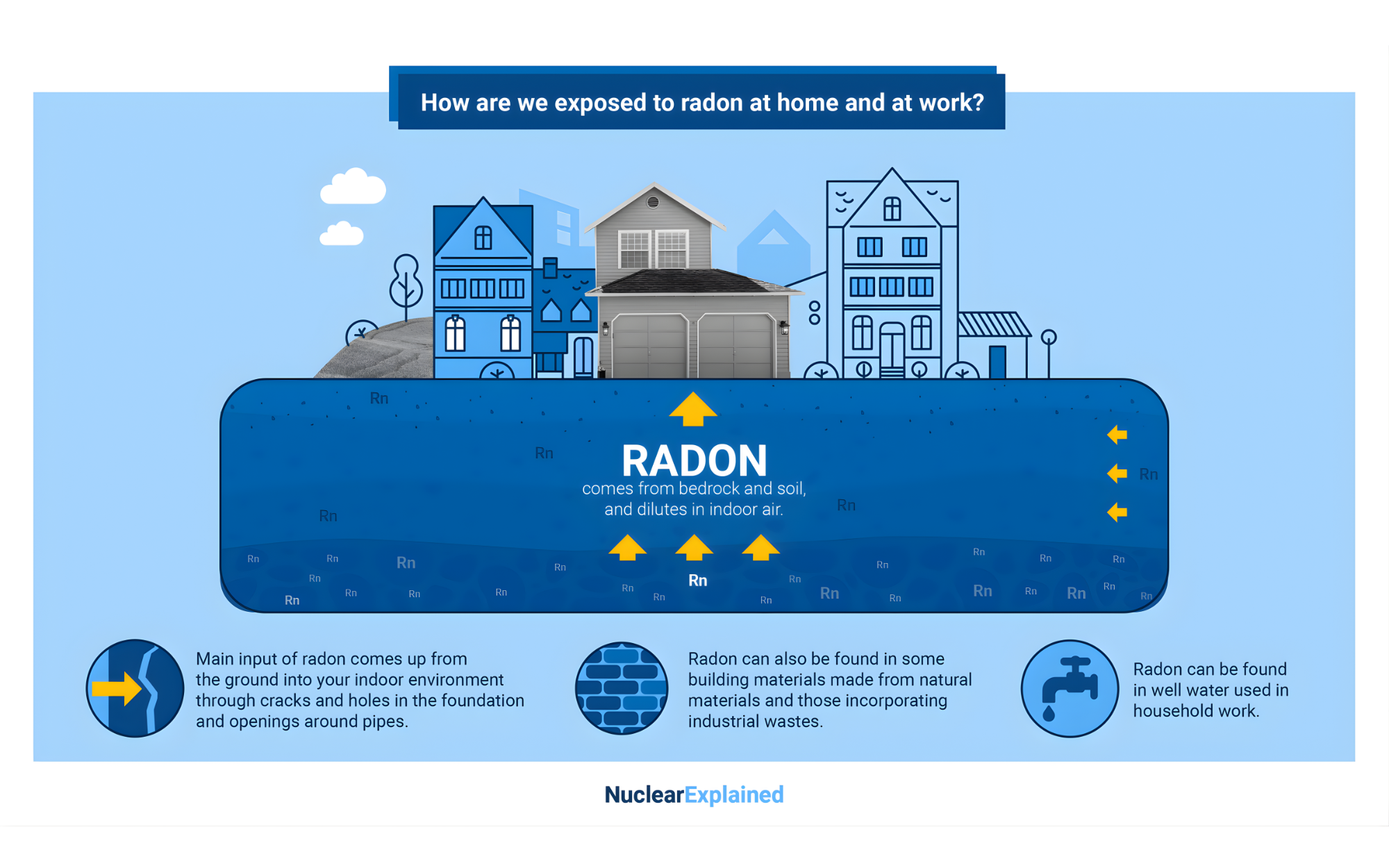
Illustration from: www.iaea.org
Mechanism of Radon Entry into Homes
Radon gas is released from the ground and can accumulate in buildings, particularly in confined areas such as basements and crawl spaces. The mechanisms of radon entry include:
Soil Gas Infiltration: The primary entry route is through the soil gas infiltration. Differences in air pressure between the inside of a building and the soil around it can create a vacuum effect, drawing radon in through cracks in the foundation, construction joints, and gaps around pipes.
Building Materials: Some building materials, such as concrete and granite, can emit radon, although this is typically a minor source compared to soil gas infiltration.
Water Supply: Radon can dissolve in groundwater and be released into the air when water is used for household activities like showering and cooking. Homes with well water may need specialized radon mitigation systems to address this.
Health Effects of Radon
Radon itself is chemically inert and poses no direct threat when inhaled. However, its decay products, known as radon progeny or daughters, are radioactive and can attach to dust and other particles in the air. When inhaled, these particles can become lodged in the lung tissue, irradiating the cells and potentially causing damage. Prolonged exposure to radon decay products increases the risk of lung cancer, making radon exposure the second leading cause of lung cancerafter smoking. This is why indoor air quality testing is now a standard recommendation from health organizations.
Historical Context and Modern Understanding
Early 20th Century
In the early 20th century, the health risks associated with radon were not well understood. It was primarily studied for its radioactive properties and potential therapeutic uses. The first significant health concerns arose from studies of miners, particularly uranium miners, who exhibited high rates of lung cancer due to prolonged exposure to radon and its progeny in poorly ventilated mines.
Mid to Late 20th Century
By the 1950s and 1960s, more systematic studies linked residential radon exposure to lung cancer risk. The development of sensitive detection methods allowed for more accurate measurements of radon levels in homes. The Environmental Protection Agency (EPA) in the United States and similar organizations worldwide began to establish guidelines and recommend radon testing in homes, particularly in areas with known uranium deposits or elevated risk.
Present Day
Today, radon is recognized as a significant indoor air pollutant. Testing and mitigation have become standard practices in many countries. The EPA recommends that homes be tested for radon gas and that mitigation measures be taken if levels exceed 4 pCi/L (picocuries per liter). Modern radon mitigation techniques include:
Soil Suction: Systems that draw radon from beneath the house and expel it outside.
Sealing Cracks and Openings: Reducing radon entry by sealing foundation cracks and openings.
Ventilation: Increasing ventilation to dilute indoor radon concentrations.
Water Treatment: For homes with radon in the water supply, treatment methods like aeration or activated carbon filters can be used.
These solutions are especially critical during real estate transactions, when compliance with radon guidelines is often required before closing.
Conclusion
The journey of radon gas from its discovery to our current understanding highlights significant advancements in both science and public health. While radon poses a serious health risk, particularly for lung cancer, effective radon testingand mitigation strategies can significantly reduce this risk, ensuring safer indoor environments. Continuous research, regulatory action, and public awareness remain crucial in managing radon exposure and protecting homeowners, real estate clients, and families alike.
